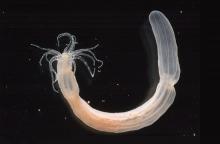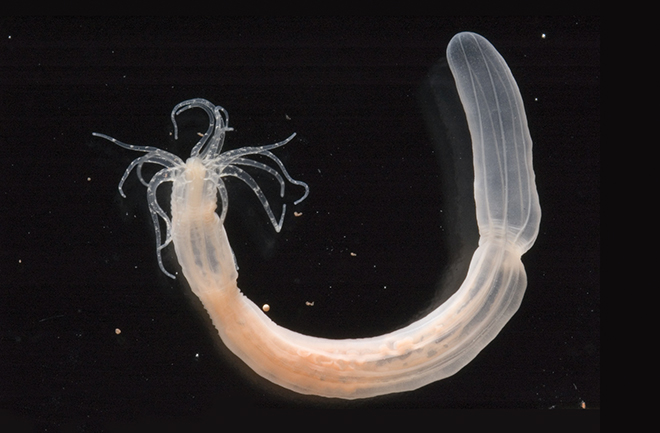
The starlet sea anemone, Nematostella vectensis, is a species of small sea anemones that occupy an essential position in the tree of life. They are primitive animals with epithelial cells, neurons, stem cells, complex extra-cellular matrix, muscle fibers, and they have extensive regenerative ability. Although the Nematostella is a “primitive” animal, the sea anemone genome is more closely related to the human genome than the current invertebrate animals used to model human biology. Understanding the neuroregenerative properties  of Nematostella is the current interest of biologist Michael Layden.
of Nematostella is the current interest of biologist Michael Layden.
Layden’s research focuses on understanding how the mechanisms that regulate regeneration of the anemone’s nervous system may vary from mechanisms that generate the nervous system during development.
“Traditionally, people have looked at the process of neurogenesis during development (when a fertilized egg develops into an adult animal), but now the goal is to understand the same process during regeneration of adult bodies,” says Layden, assistant professor of biological sciences. “There is a clear indication when animals regenerate, the mechanisms are similar to those used during development. You don’t reinvent the wheel to regenerate a limb for example, but regeneration does not use the exact same molecular program as development. In order to understand the significance of differences observed between development and regeneration, you need to study them in the same animal.”
Most current research on regeneration uses model systems that are not suited for investigating both animal regeneration and development. It is difficult to compare the neural development and neural regeneration directly, because they are investigated in two different animals, says Layden. Nematostella is one of the few animal models that allow researchers to easily access and manipulate gene function during both development and regeneration. Understanding the similarities and differences in how developmental programs are redeployed during regeneration will provide researchers with critical clues needed to design better regenerative therapies for human neurological disorders.
Layden examines achaete-scute homologs (ash), a type of gene that regulates formation of a subset of the Nematostella nervous system during embyrogenesis. He and his team disrupt this gene’s function and analyze neural phenotypes at the Nematastella’s larval and juvenile polyp stages. In conjunction with this, he is improving methods to disrupt genetic function during regeneration. The goal is to assess whether this ash gene regulates formation of the same neurons that it does during development, regulates neural development using the exact same molecular program, and/or regulates distinct neuron sub-types that are not regulated by this gene during development.
Using Nematostella allows researchers have access to the neural processes in an invertebrate model that is inexpensive to study when probing regenerative properties. Layden can obtain thousands of embryos daily and his team can dissect anemones at any stage of their lives, including during regeneration. By examining sea anemones, he and his colleagues are laying the groundwork for future studies in vertebrates, such as zebrafish and mice.
Additionally, because Nematostella is a primitive animal, Layden’s research will also provide insight about molecular programs that regulated formation of the ancestral nervous systems that gave rise to more complex nervous systems, such as the human brain.