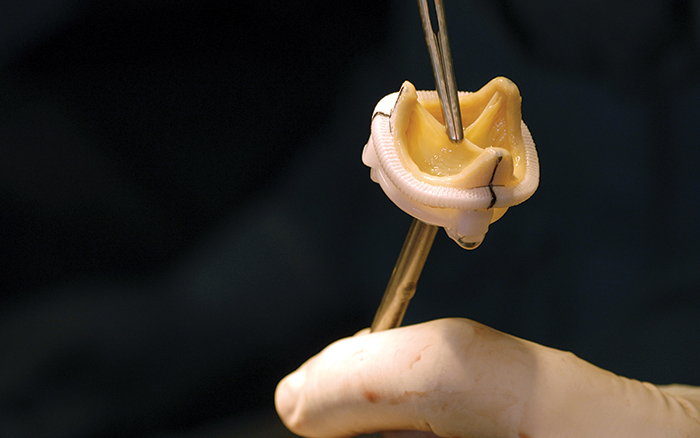
Heart valves regulate the flow of blood through the heart’s four chambers—two small, round upper chambers (atria) and two larger, cone-shaped lower chambers (ventricles). Each ventricle has a one-way “in” valve and a one-way “out” valve. Each valve consists of flaps that open and close like one-way swinging doors. When these valves become diseased, surgical repair is required, and mathematician Yue Yu has received a CAREER grant from the National Science Foundation to develop modeling of the damage process of the replacement heart valve after valve replacement.
Yu, assistant professor of mathematics, is exploring a new mathematical framework and the corresponding high-performance open source software to address fundamental questions on modeling the soft tissue damage process after heart valve replacement, with the ultimate goal of improving the understanding of heart valve damage processes.
There are two types of commonly used replacement heart valves. One is mechanical, but the drawback is that the valve is not flexible. Mechanical valves are more durable than the alternative bioprosthetic valves; however, the need for long-term anticoagulant therapy renders them unsuitable for some patients. The challenge for doctors lies with the damage to red blood cells created by this valve. Patients who receive a manufactured valve will nearly always require a blood-thinning medication throughout the remainder of their lives. The blood thinner will keep clots from forming, which is critical for the person with a mechanical valve because clots can lodge in the valve flaps or hinges and cause a malfunction. The more popular option is the bioprosthetic valves, which are created from animal donors’ valves or other animal tissue that’s strong and flexible. Tissue valves can last 10 to 20 years, and usually don’t require the long-term use of medication. For a young person with a tissue valve replacement, the need for additional surgery or another valve replacement later in life is highly likely. Because the process occurs over an extended period of time, scientists cannot easily observe the degradation.
“The damage occurs slowly, so we can’t easily observe what is happening to the valve,” says Yu. “Even after using bioprosthetic heart valves for many years, we still don’t know why the valve becomes fatigued and the damage happens. We want to develop a mathematical model that will help scientists observe what is going on over time and to make long-term predictions of what will occur over the 10 to 15 years a valve degrades.”
In the end, Yu envisions a computational approach that simulates the physically realistic process with the objective of understanding tissue fatigue mechanisms. She will develop open source software, which not only will contribute to advance the knowledge of soft tissue damage, but also will increase the variety of capabilities for computer simulation of problems in long-term materials and manufacturing damage progression in general. The computational tool and the methodology developed in this project can also be used efficiently in a wide range of physical and biological problems. Her findings will have potential impacts on heart valve operation and research and will potentially contribute toward more rigorous interventional and surgical procedures.